Measles Recurrence in a Refugee-Hosting Setting: An Outbreak Investigation in Kamwenge District, Uganda, 2025
Authors: Nasif Matovu¹*, Kyomugisha D. Aman1, Deborah Aujo1, Yasin Nuwamanya¹, Richard Migisha¹, Benon Kwesiga¹, Fred Nsubuga2 Institutional affiliations: ¹Uganda Public Health Fellowship Program, Ministry of Health, Kampala, Uganda, 2Uganda National Expanded Program on Immunisation, Ministry of Health, Kampala, Uganda Correspondence*: Tel: +256774755494, Email: matovan5@uniph.go.ug
Summary
Background: Measles remains a major cause of vaccine-preventable morbidity in Uganda, with 61/146 districts reporting a confirmed measles outbreak between January and October 2025, despite the availability of an effective Measles-Rubella (MR) vaccine. On 24 June 2025, Kamwenge, a refugee-hosting district, reported three confirmed measles cases. This was the 3rd measles outbreak in two years. We investigated to determine outbreak magnitude, identify risk factors, estimate vaccine effectiveness (VE), and vaccine coverage (VC), and recommend control prevention measures.
Methods: We defined a suspected case as acute onset of fever and generalized maculopapular rash plus ≥ 1of cough, coryza, or conjunctivitis in a Kamwenge resident during 1 April–7 July 2025; a confirmed case was a suspected case with measles IgM-positive. We identified cases through health facility records and community case search. We also conducted environmental assessments. We described cases by person, place, and time. Vaccination status was verified by the child\s health card or caregiver recall. Attack rates (AR) were calculated based on district population projections. We conducted a 1:3 age-and subcounty-matched case-control study and used conditional logistic regression to generate adjusted odds ratios (aOR). VE was calculated as (1–ORM-H) ×100. We calculated VC as the proportion of vaccinated controls.
Results: We identified 109 cases (3 confirmed, 106 suspected); no deaths, 48.6% (53/109) were refugees. Overall AR was 3.2/10,000, highest among children 9-17 months (31/10,000) and refugees (13.2/10,000). Inadequate triage and delayed isolation were observed. Visiting places of worship significantly increased odds of infection (aOR=9.9, 95% CI:3.2-30.1) while receiving one MR dose reduced odds (aOR=0.11, CI:0.03-0.48), with greater protection for two doses (aOR=0.04, CI:0.01-0.29). VE for MR1 was 92% (CI: 76-98) and 96% (CI:80-99) for MR2. VC was 67% for MR1 and 36% for MR2 among children ≥9 months.
Conclusion: The outbreak was associated with suboptimal MR coverage, mass gatherings, and inadequate triage. A mass vaccination campaign (6-59 months) and strengthened isolation of febrile rash cases were implemented.
Background
The measles virus causes a severe systemic illness (1). Transmission of Measles is dependent on person-to-person spread through respiratory droplets or direct contact (2). Measles has an incubation period of approximately 10-14 days, a basic reproductive number (R₀) ranging from 12 to 18, making it one of the most contagious human diseases, with a case fatality rate of 1-3% in low-resource settings (3). Furthermore, measles virus infection diminishes preexisting antibodies that offer protection from other pathogens (4). Measles is re-emerging in several developed countries despite having an effective vaccine for decades. This resurgence is due to insufficient vaccination coverage in certain subpopulations, the anti-vaccination movement, and increased human mobility across borders(5). In Uganda, recurrent measles outbreaks continue to occur due to suboptimal routine immunization coverage, delayed supplemental immunization activities, and immunity gaps in refugee-hosting and hard-to-reach populations(6-8).
On June 24, 2025, Uganda’s Ministry of Health reported a measles outbreak in Kamwenge District, with three of five samples testing positive for measles-specific IgM antibodies. The district has faced ongoing outbreaks due to low immunization rates, immunity gaps, and challenges in accessing refugee and hard-to-reach populations, raising concerns about local transmission and preparedness(6-8). We assessed the outbreak’s scope, response timeliness, transmission risk factors, vaccine effectiveness, coverage, and recommended control measures.
Methods
The measles outbreak occurred in Kamwenge, a district in Western Uganda with a population of 337,167, including 39,929 refugees across 17 sub-counties. Kamwenge has a history of measles outbreaks, including one in February 2025, where 75% of case-patients had never received any Measles-Rubella vaccinations. The outbreak lasted from April to July 2025, affecting select sub-counties.
We conducted descriptive epidemiology to understand the distribution of cases by person, place, and time
We conducted an environmental assessment to identify factors that facilitated measles transmission at Rwamwanja Health Center IV, the main measles treatment unit, and within affected communities to identify transmission risks, such as crowded settings, including schools and places of worship.
We conducted thirty-one hypothesis-generating interviews with parents or guardians of case-patients to assess exposures during the three weeks before symptom onset.
We conducted an age-matched case-control study in a ratio of 1: 3 controls to test the generated hypotheses, with controls selected from children without signs and symptoms suggestive of measles. Vaccination status was confirmed through cards or caregiver recall.
We estimated vaccine effectiveness using the formula VE = (1 – ORM-H), while the vaccine coverage was estimated using the proportion of eligible vaccinated respondents among the control group.
Whole blood samples were collected from suspected case-patients for measles IgM antibody testing using enzyme-linked immunosorbent assay (ELISA) kits.
This outbreak investigation was conducted as a public health response and classified as non-research. Approval was obtained from the Ministry of Health (MoH). The investigation was also approved by the US CDC and conducted in accordance with the applicable US federal laws, and permission was granted by the Kamwenge District Health Office. Verbal informed consent was obtained from respondents, and confidentiality was maintained throughout the investigation.
Results
Descriptive epidemiology
We recorded 109 measles cases, including three confirmed and 106 suspected. Seventy-nine percent (86/109) had fully recovered, with no deaths. Fifty-three percent were refugees, with an overall attack rate (AR) of 3.2/10,000, affecting females more (AR 3.7/10,000) than males (AR 2.8/10,000). The most affected age group was 9-17 months AR (30.8/10,000), followed by 18-59 months AR (7.7/10,000). Refugees had a higher AR of 13.3/10,000 compared to nationals at AR (1.9/10,000). All cases reported fever and maculopapular rash, with many also experiencing coryza (71%), cough (61%), and conjunctivitis (54%).
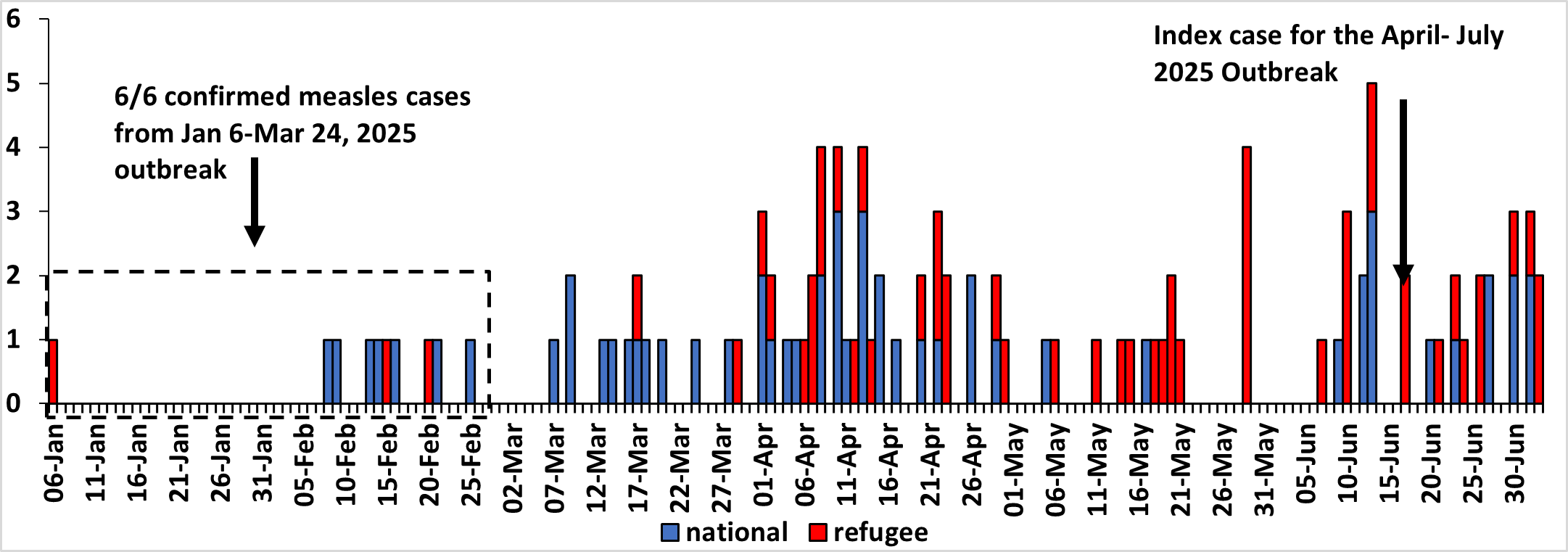
The index case for the April-July outbreak developed a rash on the 17th of June 2025. However, following active case search, we identified suspected cases dating as far back as 6th January (Figure 2).
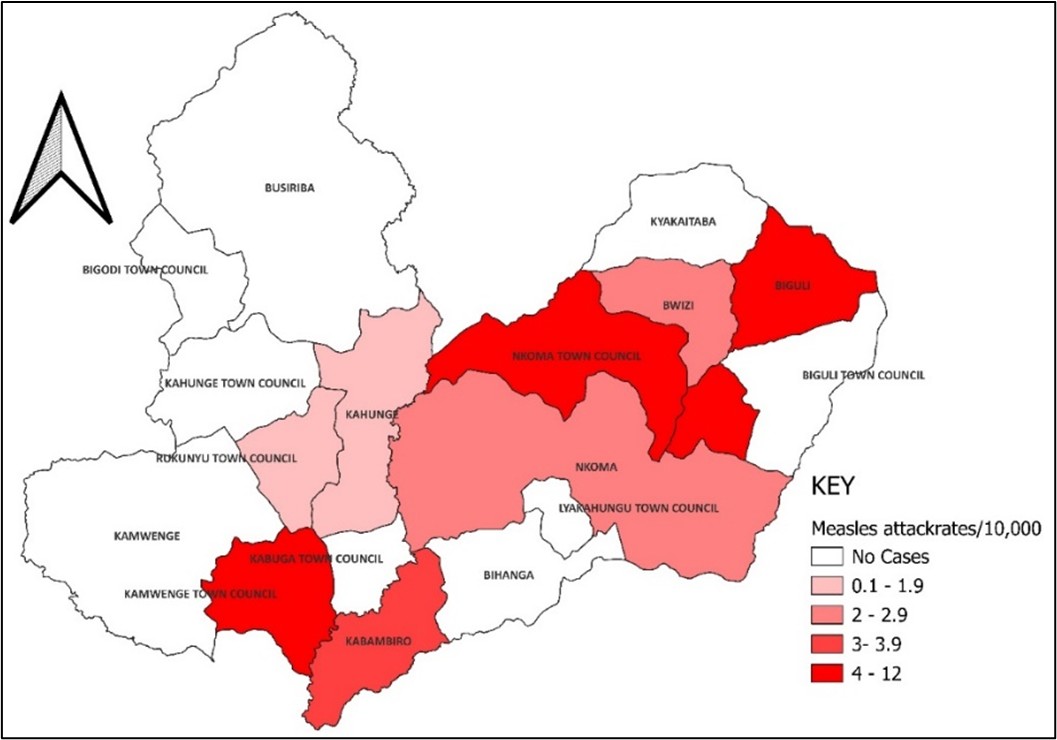
The overall attack rate was 3.2/10,000 with subcounties of Nkoma Katalyeba Town Council (AR:12/10,000), Biguli subcounty (AR:8/10,000) reporting the highest attack rates (Figure 3).
Environmental assessment findings
The assessment found that measles patients were mixed with other pediatric cases at Rwamwanja HCIV and Biguli HC III. There were no functional isolation areas and inadequate triage processes. Large gatherings in Nkoma Katalyeba, combined with a lack of awareness about measles transmission, contributed to the spread of the infection.
Hypothesis generation findings
Table 1: Summary of the risk factors for Measles transmission in Kamwenge district
| Risk factor | Percentage % |
| Attending a social gathering | 79% |
| Attending a place of worship | 66% |
| Attending school within 3 weeks before falling sick | 53% |
| Not vaccinated against measles | 46% |
| Visiting a water collection point | 39% |
| Playing away from home | 32% |
| Attending a health facility | 25% |
The potential risk factors for measles transmission included attending social gatherings (especially places of worship), going to school, and being unvaccinated (Table 1).
Case-control study findings
Children who received at least one dose of the measles-rubella (MR) vaccine had 89% lower odds of contracting measles (aOR 0.11; 95% CI 0.032-0.48), and those with at least two doses experienced a 96% reduction in risk (aOR 0.04; 95% CI 0.01-0.29). Additionally, visiting a place of worship three weeks before symptom onset increased the odds of measles infection by 10-fold (aOR 9.5; 95% CI 3.6-25) (Table 2)
Table 2: Risk factors for measles transmission, Kamwenge, April– July, 2025
| Risk factor* | Number (%Exposed) | cOR (95%CI)
|
aOR (95%CI) | |
| Cases
n (%) |
Controls
n (%) |
|||
| One Measles-Rubella vaccine dose | ||||
| Received | 32(50) | 97(86) | 0.08(0.22-0.29) | 0.11(0.03-0.48) |
| Did not receive | 32(50) | 16(14) | Ref | |
| Two Measles-Rubella vaccine doses | ||||
| Received | 9(22) | 54(77) | 0.04(0.01-0.21) | 0.04(0.01-0.29) |
| Did not receive | 32(78) | 16(23) | Ref | |
| Attending a place of worship | ||||
| Attended | 40(55) | 30(18) | 9.9(3.9-25) | 9.5(3.6-25) |
| Did not attend | 33(45) | 137(82) | ||
We also assessed the relationship between becoming a case and the visiting a water collection point, school attendance, playing away from home, history of travel away from home and visiting a health facility before developing a rash and these were not significant.
Measles vaccine effectiveness
In the case-control analysis, 50% (32/64) of cases had received one dose of the measles vaccine, which was 93% effective against infection (ORMH 0.07; 95% CI=0.02-0.23). Overall, the vaccine effectiveness for one dose was estimated at 93% (95% CI=77-98), while those who received a second dose had even higher effectiveness at 95% (95% CI=78-99).
Measles vaccination coverage
Sixty-seven percent (105/156) of controls aged at least 9 months received at least one dose of the measles vaccine. Vaccination coverage was highest among children aged 9-17 months. Nationals had a higher coverage at 71%, while refugees were at 66%. The coverage for the second dose of MR2 was lower at 36%, with nationals only at 8% compared to refugees at 49%.
Discussion
Low immunization coverage and high-risk social mixing contributed to the April–July 2025 measles outbreak in Kamwenge District, consistent with patterns observed in Uganda and across the African Region. Recent outbreaks in districts such as Terego and Kakumiro have similarly highlighted persistent gaps, particularly among displaced populations where MR2 uptake remains low and susceptible populations accumulate over time(6-8). The higher attack rates observed among refugees in this investigation further underscore the vulnerability of displaced populations when routine immunization services are inconsistently accessed.
Crowded indoor gatherings, particularly religious congregations, likely amplified transmission. Evidence from outbreak investigations indicates that even small declines in vaccination rates can trigger rapid outbreaks in these settings, due to intense social mixing(10). In this study, unvaccinated children attending places of worship had significantly higher odds of measles infection, consistent with previous Ugandan outbreak investigations identifying crowded social environments as key drivers of transmission. (11, 12)
Vaccination provided strong protection, with vaccine effectiveness estimated at 93% for one dose and 95% for two doses, consistent with field investigations and controlled trials (13). However, coverage remained suboptimal, with MR1 coverage at 67% and MR2 coverage at 36%, both below the ≥95% threshold required to interrupt transmission. Lower MR2 coverage and higher attack rates among refugee populations highlight the need for targeted strategies to improve completion of the two-dose schedule.
Health-facility factors also contributed to transmission. Mixing suspected measles cases with other patients and the absence of functional triage at Rwamwanja HC IV likely facilitated nosocomial transmission, as observed in other Ugandan measles investigations. (8) Strengthening infection prevention measures, including effective triage, isolation areas, and improved outpatient patient flow, is essential during outbreaks.
Globally, measles is considered a key indicator of immunization program performance, and WHO recommends achieving and sustaining≥95% coverage with two doses of measles- containing vaccine. Global strategies, including the Measles & Rubella Strategic Framework and Immunization Agenda 2030, emphasize closing immunity gaps through strengthened routine immunizations, targeted supplemental activities (SIAs), and strategies for underserved populations(14, 15). The findings from Kamwenge Support these priorities
Study limitations
Vaccination status partly relied on caregiver recall, where cards were unavailable, which may introduce recall bias. Coverage estimates based on controls from affected sub-counties, which might not reflect the entire district, and the small number of laboratory samples tested may have underestimated confirmed cases.
Public health actions
A mass vaccination campaign targeting all children aged 6 to 59 months was conducted in all sub-counties. Active case finding was intensified in health facilities and communities, and triage and temporary holding areas were set up in outpatient departments. Social mobilization and risk communication efforts were also enhanced.
Conclusion
The outbreak was driven by immunity gaps from low MR1 and MR2 coverage, amplified by high-risk social mixing and gaps in infection prevention in health facilities. High vaccine effectiveness indicates that programmatic weakness in vaccination delivery, rather than vaccine failure were the primary driver.
Recommendations
Routine immunization should be strengthened to achieve and sustain ≥95% MR1 and MR2 coverage through targeted outreach and defaulter tracking. SIAs should prioritize refugee settlements and underserved communities. Health facilities should improve triage, isolation, and infection prevention practices, while community-based surveillance and engagement with religious and community leaders should be intensified to support vaccination uptake and outbreak control.
Conflict of interest: The authors declare that they have no conflict of interest
Author’s contributions
NM, KDA, and AD designed the study and contributed to the data collection and analysis. NM led the writing of the bulletin. NM YN, AD participated in bulletin writing and review to ensure scientific integrity and intellectual content. All the authors contributed to the final draft of the bulletin
Acknowledgements
We appreciate the management of Kamwenge District Local Government and Rwamwanja HC IV for their stewardship, and the community for their participation in this investigation. We also thank the Ministry of Health, Uganda, National Expanded Program for Immunisation (UNEPI) for their technical support. We are grateful to the Global Alliance for Vaccines and Immunisation (GAVI) and the US CDC for funding this investigation.
Copyright and licensing
All materials in the Uganda Public Health Bulletin are in the public domain and may be used and reprinted without permission. However, citation as to source is appreciated. Any article can be reprinted or republished. If cited as a reprint, it should be referenced in the original form.
References
- Griffin DE, Ward BJ, Esolen LM. Pathogenesis Of Measles Virus Infection: An Hypothesis For Altered Immune Responses. The Journal of Infectious Diseases. 1994;170(Supplement_1):S24-S31.
- Bester JC. Measles and Measles Vaccination: A Review. JAMA Pediatrics. 2016;170(12):1209-15.
- Organisation WH. Measles Fact Sheet Genever: WHO; 2023 [cited 2025 04/10/2025]. Available from: https://www.who.int/news-room/fact-sheets/detail/measles.
- Mina MJ, Kula T, Leng Y, Li M, de Vries RD, Knip M, et al. Measles virus infection diminishes preexisting antibodies that offer protection from other pathogens. Science. 2019;366(6465):599-606.
- Rota JS, Hickman CJ, Sowers SB, Rota PA, Mercader S, Bellini WJ. Two Case Studies of Modified Measles in Vaccinated Physicians Exposed to Primary Measles Cases: High Risk of Infection But Low Risk of Transmission. The Journal of Infectious Diseases. 2011;204(suppl_1):S559-S63.
- Okiror Okello E, Migisha R, Ampaire I, Nsubuga F, Nalwanga J, Kwizera P, et al. Measles outbreak investigation in Kakumiro District, Uganda, February–May 2024. Discover Public Health. 2025;22(1):1-10.
- Namusisi AM, Nuwamanya Y, Migisha R, Atugonza R, Immaculate A, Nsubuga F, et al. Measles outbreak Investigation in Terego District, Uganda, May-June, 2024.
- Kizito SN, Simbwa B, Thomas K, Akuguzibwe R, Sembatya I, Nsubuga E, et al. Measles outbreak propagated by visiting a health facility, in a refugee hosting community, Kiryandongo District, Western Uganda, August 2022–May 2023.
- Nsubuga F, Bulage L, Ampeire I, Matovu JK, Kasasa S, Tanifum P, et al. Factors contributing to measles transmission during an outbreak in Kamwenge District, Western Uganda, April to August 2015. BMC infectious diseases. 2018;18(1):21.
- Measles, Initiative R. Perspective: The risks of measles in religious communities. Retrieved October. 2013;30:2013.
- Namulondo E, Ssemanda I, Komugisha M, Nuwamanya Y, Nsubuga EJ, Wako S, et al. Measles Outbreak in a Refugee Settlement, Kiryandongo District, Uganda, July–October 2023. 2024.
- Majwala RK, Nakiire L, Kadobera D, Ario AR, Kusiima J, Atuhairwe JA, et al. Measles outbreak propagated by children congregating at water collection points in Mayuge District, eastern Uganda, July–October, 2016. BMC infectious diseases. 2018;18(1):412.
- CDC. Measles Vaccination 2025 [cited 2025 24/08/2025]. Available from: https://www.cdc.gov/measles/vaccines/index.html.
- WHO. Immunization Agenda 2030 2020 [cited 2025 27092025]. Available from: https://www.who.int/publications/m/item/immunization-agenda-2030-a-global-strategy-to-leave-no-one-behind.
- WHO. Measles Outbreak Toolkit 2022 [cited 2025 01102025]. Available from: https://www.who.int/emergencies/outbreak-toolkit/disease-outbreak-toolboxes/measles-outbreak-toolbox.


Comments are closed.