Clustered deaths linked to severe malaria and poor health-seeking behavior in Mid-Western Uganda, June–September 2025
Authors: Michael Mutegeki¹*, Nasif Matovu¹, Aman Denise Kyomugisha¹, Florence Nambaziira¹, Vivian Nakaweesa¹, Peter Chris Kawungezi², Atek Kagirita³, Benon Kwesiga¹, Richard Migisha¹ Institutional affiliations: ¹Uganda Public Health Fellowship Program, Uganda National Institute of Public Health, Kampala, Uganda, ²Mubende Regional Referral Hospital, Mubende, Uganda, ³Department of Integrated Epidemiology, Surveillance and Public Health Emergencies, Ministry of Health, Kampala, Uganda Correspondence*: Tel:+256787882985, Email: mutegekim@uniph.go.ug
Summary
Background: Uganda remains one of the top 3 countries most affected by malaria with a nationwide prevalence of 19% and endemicity in over 95% of the districts. In August 2025, clusters of rapidly progressing deaths were reported in Mubende, Kakumiro, and Kyankwanzi districts in mid-western Uganda. We investigated to determine the cause, magnitude, contributing factors, to recommend public health measures to prevent more related deaths.
Methods: A suspected case was defined as an acute onset of ≥2 symptoms (fever, headache, dizziness, abdominal pain, vomiting, or weakness) in a resident of the affected sub-counties (Kibijjo, Kisala, Butoloogo and Southern Division) from June 1–September 5, 2025. Cases were identified through healthcare facility records review and active case search with house-to-house searches assisted by local leaders. We interviewed cases on demographics, symptoms, exposures, and health-seeking behavior. Samples (blood, urine, stool) were picked and tested for infectious and toxicological causes. We also analyzed malaria surveillance data. We summarized alive cases and deaths using descriptive epidemiology (proportions and attack rates by age group, sex and sub-county).
Results: We identified 57 cases from 46 households. Thirteen (23%) died, with 8/13 (62%) dying within 24 hours of symptom onset. The majority of cases were children aged 6–18 years (38/57,65%). One-third (15/46) of households had ≥1 case. Kisala Sub-county was the most affected with an attack rate of 409 cases per 100,000 population. Out of the total cases, 40/57 (70%) first sought care at drug shops/clinics and 12/57 (21%) self-medicated. Of 1,304 records reviewed, malaria was the leading diagnosis with 716 cases (55%) and a test positivity rate of 57%. Plasmodium falciparum was detected in 14/22 (64%) of the tested samples. Malaria surveillance data of the affected region showed case counts consistently exceeded the epidemic threshold from week 1–27 of 2025, confirming a malaria upsurge. All cases tested negative for viral hemorrhagic fevers and toxicological agents.
Conclusion: The clustered deaths were caused by severe malaria, exacerbated by poor health-seeking behaviors. We recommended enhancing community-based mortality surveillance, risk communication and test-and-treat approaches to reduce transmission and prevent more related deaths.
Background
Uganda remains one of the top 3 countries most affected by malaria (1) with a nationwide prevalence of approximately 19% and endemic in over 95% of the districts in the country. Unexplained clustered deaths represent a major public health concern due to their potential to signal emerging infectious diseases, outbreaks of unknown pathogens, environmental or toxicological exposures(2). Such events can rapidly overwhelm local health systems, create fear within communities, and pose significant threats to national health security if not promptly investigated(3).
Globally clustered deaths have been associated with various causes, including infectious diseases such as viral hemorrhagic fevers, severe malaria, bacterial meningitis, or anthrax; exposure to toxins such as pesticides or contaminated food; or sociocultural practices that facilitate rapid transmission of pathogens(4). In addition, clusters of unexplained deaths are associated with delayed detection of novel pathogens, as seen with Ebola virus disease (EVD) (5) and Covid-19, underscoring the need for timely and systematic investigation.
On August 7, 2025, the Mubende District Surveillance Focal Person (DSFP) reported a cluster of unexplained deaths to the Mubende Regional Public Health Emergency Operations Centre (RPHEOC). The deaths occurred within a short time frame and were characterized as an acute illness with high fatality, raising suspicion of a possible outbreak or exposure to a common risk factor. Preliminary verification by the RPHEOC confirmed additional clusters of deaths in neighboring Kyankwanzi and Kakumiro districts. We investigated to establish the cause of the clustered deaths, assess the magnitude of the illness, identify factors contributing to the deaths, and recommend evidence-based interventions to prevent further spread and improve preparedness for similar future events.
Methods
We investigated in Mubende, Kyankwanzi, and Kakumiro districts in mid-Western Uganda. This districts had experienced an EVD outbreak in 2022. These districts are characterized by scattered rural settlements, subsistence farming, and limited access to health services.
We defined a suspected case as; an acute onset of ≥2 of the following symptoms; headache, general body weakness, fever, dizziness, vomiting, and abdominal pain in a resident of the affected sub-counties (Kibijjo, Kisala, Butoloogo and Southern Division) of Kyankwanzi, Mubende, and Kakumiro districts from June 1, 2025 to September 5, 2025.
We reviewed symptoms from the preliminary reports and then used a structured case investigation form to conduct active case search. We conducted a house to house search starting with households that had reported deaths since June 1, 2025 and then included all their neighborhood households that had cases. We interviewed all alive cases and caretakers of cases who had passed on demographics, symptoms onset, health seeking behaviors, and outcomes.
We reviewed records (outpatient and inpatient registers) of selected high volume health facilities serving the affected sub-counties covering the June 1 to September 5, 2025.
We analyzed malaria data from District Health Information System 2 (DHIS2) for the affected districts and constructed malaria surveillance channels. We also assessed the trend of inpatient malaria cases from January to July 2025 using the Mann–Kendall trend test.
We collected a total of 86 samples from 44 alive case-patients. They were analyzed for malaria parasites, metagenomics sequencing, pathogen discovery and for viral hemorrhagic fever/arbovirus using Polymerase Chain Reaction (PCR) and toxicology tests done.
We entered all collected data into a standardized Kobo Collect questionnaire. We conducted descriptive epidemiology on the identified cases.
We conducted the investigation in response to a public health emergency as part of the National Rapid Response Team of the Ministry of Health in Uganda as non-research. Verbal informed consent was obtained from cases or caretakers as proxy for minors or cases that had died prior to the start of each interview.
Results
Descriptive epidemiology
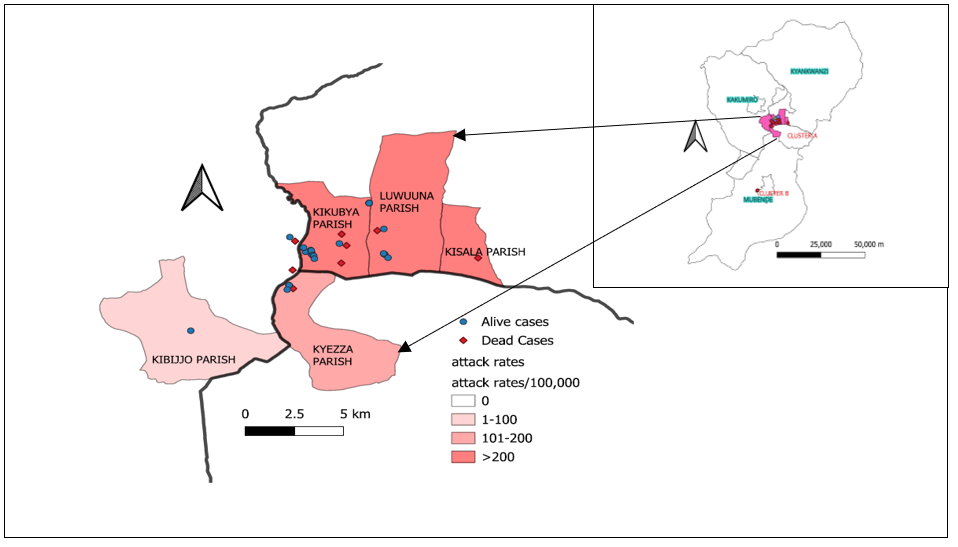
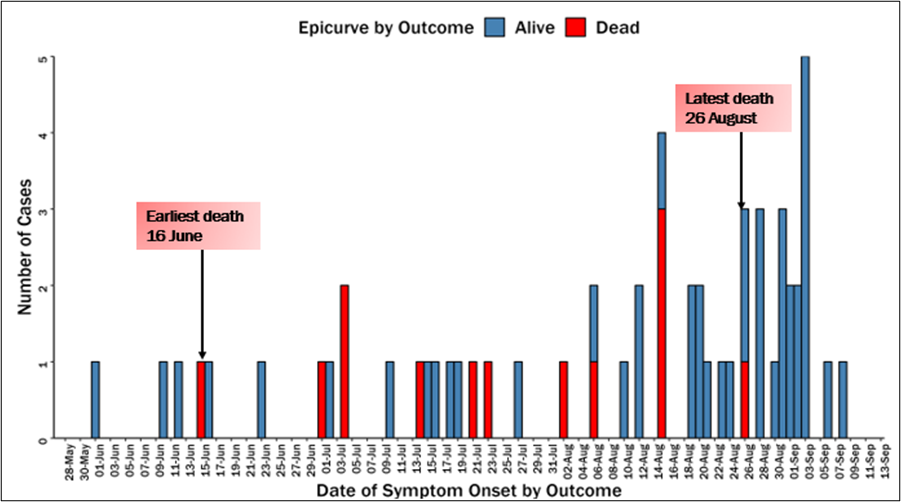
We identified a total of 57 cases. 13/57 had died yielding an overall case fatality rate of 23% with 8/13 (62%) dying within the first 24 hours of symptom onset. Children and adolescents aged 6–18 years were the most affected with attack rates 10 times higher than that of adults >18 years. Kisala was the most affected sub-county (Table 1).
Table 1: Attack rates by sex, age and Sub-counties during Clustered deaths investigation in Mubende region, June–August 2025
| Characteristics | Cases | Percentage (%) | Population | Attack rate
(per 100,000) |
| Sex | ||||
| Male
Female |
23
34 |
40.4
59.6 |
590,964
637,659 |
3.9
5.3 |
| Age group (Years) | ||||
| 0-5
6 -13 13-18 ≥ 18 Sub-counties Butoloogo Kibijo Kisala Southern Division |
15
23 15 4
15 1 38 3 |
26
41 26 7
26 2 67 5 |
206,451
258,724 200,527 613,618
32,307 30,525 9,284 42,789 |
7.8
8.9 7.5 0.7
46 3.3 409 7.0 |
We identified two clusters A and B with cluster A having 95% (54/57) of cases (Figure 1)
Cases were uniformly distributed over the three months (not clustered in time) (Figure 2).
Health seeking behaviors
We found 28/57 (67%) cases sought care first from a private clinic and 13/57 (24%) of cases opted for self-medication at home. Further analysis revealed that over half of the cases, 32/57 (56%) sought medical attention more than 24 hours after symptom onset.
Records review results
We reviewed a total of 1,304 patient records from 5 high volume health facilities in the affected districts. Malaria was the most common diagnosis 717/1,304 (55%) with an overall test positivity rate of 57%.
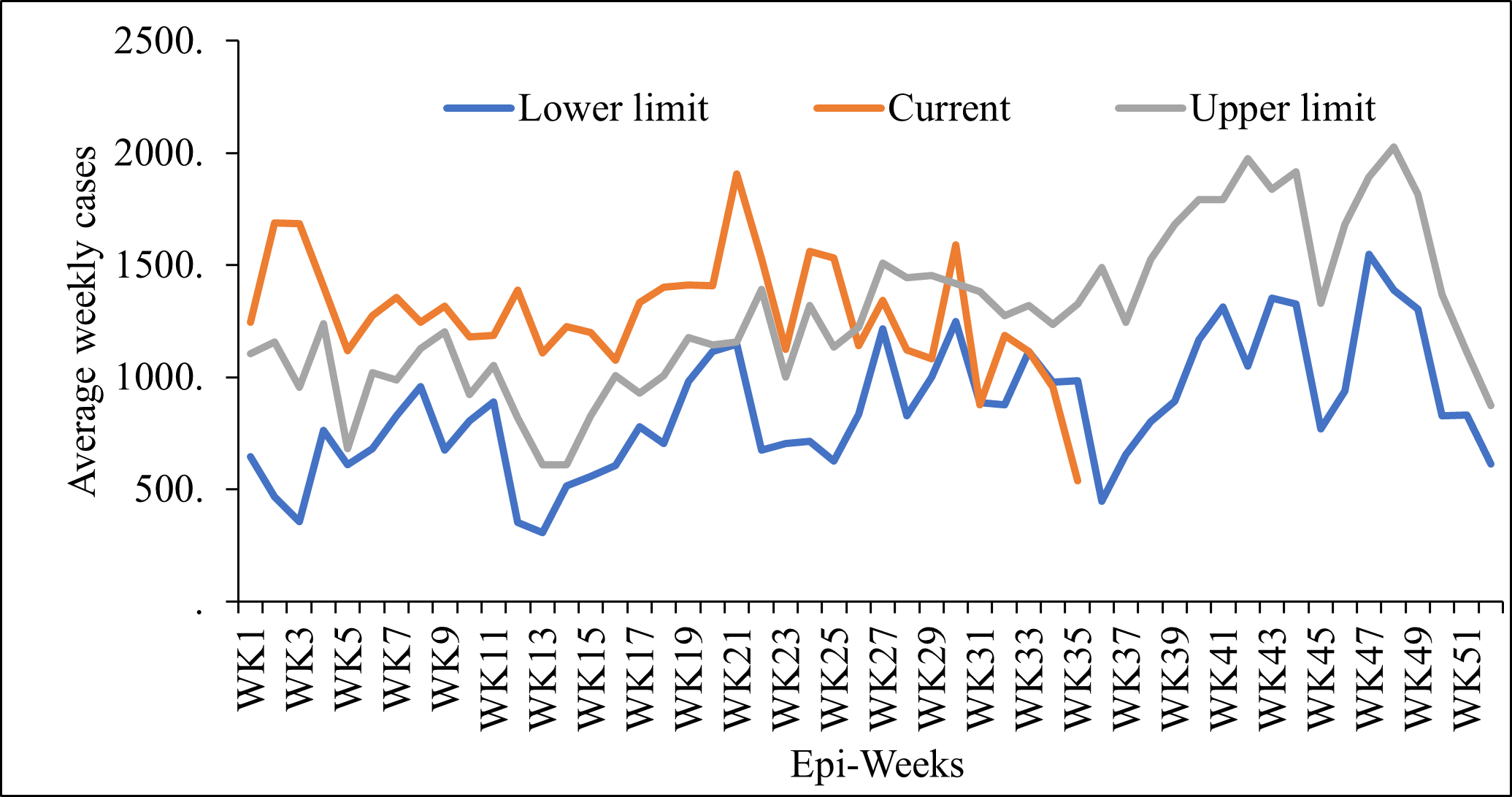
Results from analysis of District Health Information System 2 data for the affected districts
From the DHIS2 analysis, weekly surveillance data for Mubende District revealed that the number of malaria cases remained persistently above the upper limit of the normal malaria threshold for most weeks of the year (Figure 3). For Kakumiro and Kyankwanzi districts, the cases were within the normal malaria channels.
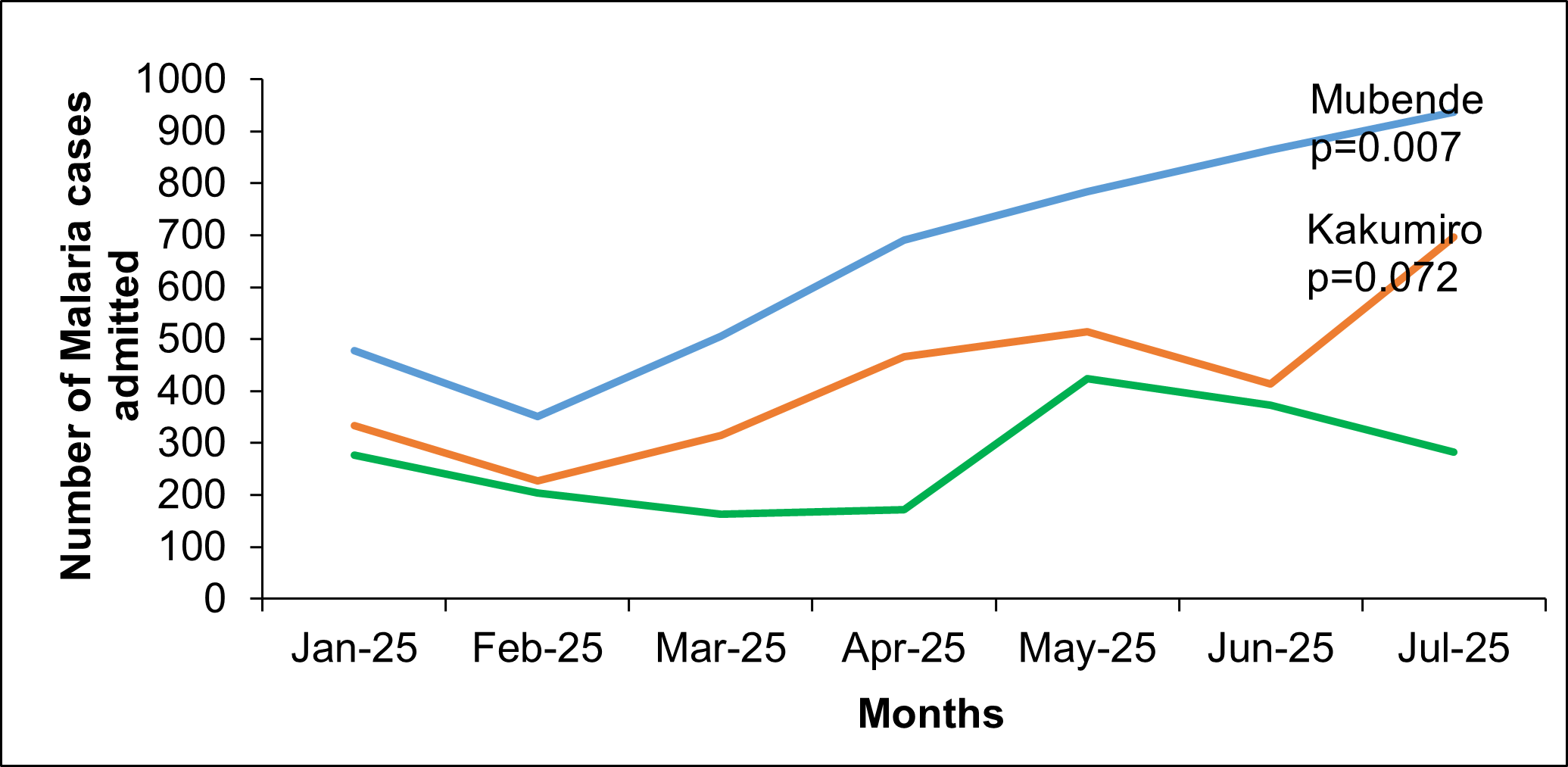
Additionally, analysis of inpatient malaria admissions also demonstrated a progressive increase across two of the three affected districts from January to July 2025. A statistically significant upward trend was observed for Mubende (p=0.007), whereas the changes in Kakumiro and Kyankwanzi were not statistically significant (Figure 4).
Laboratory investigations
Out of the 22 samples tested by PCR, 14/22 (64%) were positive for Plasmodium falciparum with 7/14 (50%) having high parasitemia. All samples tested negative for viral hemorrhagic fevers and arboviruses. Toxicological analysis similarly tested negative for heavy metals and there was no bacterial growth from blood and stool cultures.
Discussion
The investigation revealed that the clustered deaths in Mubende, Kyankwanzi, and Kakumiro districts were due to severe malaria, against a backdrop of poor health-seeking behavior. Most deaths occurred within 24 hours of illness onset is consistent with previous literature documenting the rapid progression of severe Plasmodium falciparum malaria, particularly if untreated (6).
Children below 18 years were most affected. This aligns with known malaria epidemiology, where children are most vulnerable due to low acquired immunity (7).
The epidemiological curve indicated sustained transmission from June to August 2025.This temporal pattern is consistent with seasonal malaria transmission in mid-western Uganda, where peaks coincide with the rainy season (8).
The investigation revealed delayed and inappropriate health-seeking behaviors greatly contributing to the high fatality rate. Such delays increase the risk of severe malaria and death. This is consistent with other studies done in Uganda about delayed time to seek healthcare (9).
The laboratory results strongly support malaria as the etiology, with 64% PCR positivity for P. falciparum and confirmation of high parasitemia in several cases. Importantly, the exclusion of VHFs, bacterial infections, and toxic exposures strengthens the attribution of clustered deaths to malaria. This contrasts with some previous unexplained death clusters in Uganda that were later linked to zoonotic or toxicological etiologies reinforcing the importance of comprehensive differential testing(10).
Study limitations
During this investigation, we were unable to obtain biological samples from the deceased cases because they had already been buried by the time the team arrived. This could have introduced a misclassification bias since we relied on caregiver reports to reconstruct the course of illness. Additionally, the investigation relied on ability of the participant to recall, introducing potential recall bias regarding onset of symptoms, care-seeking behavior, and exposures. Despite these limitations, the investigation provides critical insights into the pattern, context, and possible drivers of the clustered deaths and offers evidence to inform targeted public-health action.
Conclusion
Our results demonstrate that clustered deaths in Mubende, Kakumiro, and Kyankwanzi districts were most plausibly attributable to severe malaria among school-aged children, exacerbated by poor health seeking behavior.
Public health actions
We identified and treated all symptomatic cases in the community and initiated VHT-led follow-up and referral of severe cases. Additionally, we held district-led community dialogues and health education to address myths and cultural barriers. Through the district Health Teams, we quantified and redistributed Long Lasting Insecticidal Nets to the affected Sub-counties.
Recommendations
We recommended enhancing community-based mortality surveillance and risk communication to easily identify severe cases. Using the test-and-treat approaches could prevent related deaths.
Conflict of interest
The authors declare that they had no conflict of interests.
Authors’ contributions
MM, NM, ADK, FN, VN Conceived and designed the study, acquired, analyzed, interpreted the data and wrote the first draft of the manuscript. PCK and AK were the team leads of the investigation. RM, BK, provided technical supervision and Bulletin review.
Acknowledgements
We would like to appreciate the Uganda Public Health Fellowship Program for the technical support provided during the preparation and conducting the investigation. We appreciate the Uganda Ministry of Health (MoH), National Malaria Elimination Division, Mubende Regional Emergency Operation Centre, Mubende, Kakumiro and Kyankwanzi District health teams for supporting the outbreak response efforts.
Copyright
All materials in the Uganda Public Health Bulletin are in the public domain and may be used and reprinted without permission. However, citation as to source is appreciated. Any article can be reprinted or published. If cited as a reprint, it should be referenced in the original form.
References
- Venkatesan P. The 2023 WHO World malaria report. The Lancet Microbe. 2024 Mar;5(3): e214. doi:10.1016/S2666-5247(24)00016-8
- Hajjeh RA, Relman D, Cieslak PR, Sofair AN, Passaro D, Flood J, et al. Surveillance for Unexplained Deaths and Critical Illnesses. Emerg Infect Dis. 2002 Feb;8(2):145–53. doi:10.3201/eid0802.010165
- Wartenberg D. Investigating Disease Clusters: Why, When and How? Journal of the Royal Statistical Society Series A: Statistics in Society. 2001 Jan 1;164(1):13–22. doi:10.1111/1467-985X.00181
- Guthmann JP, Bonnet M, Ahoua L, Dantoine F, Balkan S, Van Herp M, et al. Death Rates from Malaria Epidemics, Burundi and Ethiopia. Emerg Infect Dis. 2007 Jan;13(1):140–3. doi:10.3201/eid1301.060546
- Curran KG, Gibson, JJ, MD, Marke, D, MD, Caulker V, et al. Cluster of Ebola Virus Disease Linked to a Single Funeral — Moyamba District, Sierra Leone, 2014. MMWR Morb Mortal Wkly Rep. 2016 Mar 4;65(8):202–5. doi:10.15585/mmwr.mm6508a2
- Yap XZ, McCall MBB, Sauerwein RW. Fast and fierce versus slow and smooth: Heterogeneity in immune responses to Plasmodium in the controlled human malaria infection model. Immunological Reviews. 2020 Jan;293(1):253–69. doi:10.1111/imr.12811
- Doolan DL, Dobaño C, Baird JK. Acquired Immunity to Malaria. Clin Microbiol Rev. 2009 Jan;22(1):13–36. doi:10.1128/CMR.00025-08
- Kigozi SP, Kigozi RN, Sebuguzi CM, Cano J, Rutazaana D, Opigo J, et al. Spatial-temporal patterns of malaria incidence in Uganda using HMIS data from 2015 to 2019. BMC Public Health. 2020 Dec;20(1):1913. doi:10.1186/s12889-020-10007-w
- Rutebemberwa E, Kallander K, Tomson G, Peterson S, Pariyo G. Determinants of delay in care‐seeking for febrile children in eastern Uganda. Tropical Med Int Health. 2009 Apr;14(4):472–9. doi:10.1111/j.1365-3156.2009.02237.x
- Bbosa N, Ssemwanga D, Weiss SL, Kalungi S, Mawanda A, Ssentudde R, et al. Identification of Anthrax as the Cause of a Cluster of Unexplained Deaths, Uganda, 2023: The Role of Metagenomic Next-Generation Sequencing and Postmortem Specimens. The American Journal of Tropical Medicine and Hygiene. 2025 Apr 2;112(4):835–9. doi:10.4269/ajtmh.24-0489


Comments are closed.