Effect of surge team deployment on Turnaround Time for result receipt during the Mpox outbreak, Mayuge District, Uganda, September– October 2024
Authors: Winnie Agwang1,2, Wilfred Opeli1,Tracy Rutogire1, Ritah Namusoosa1, Rogers Kisame3, Susan Nabadda4, Yusufu Kaweesi4, Richard Isabirye5, Samuel Gidudu1 Institutional affiliations: 1Uganda Public Health Fellowship Program-Laboratory Leadership Program, Uganda National Institute of Public Health, Kampala, Uganda; 2World Health Organization, Kampala, Uganda; 3Baylor College of Medicine Children’s Foundation, Kampala, Uganda; 4Uganda National Health Laboratory and Diagnostic Services, Kampala, Uganda; 5Mayuge District Local Government, Mayuge, Uganda. Correspondence*: Tel: +256 778192948, Email: wagwang@uniph.go.ug
Summary
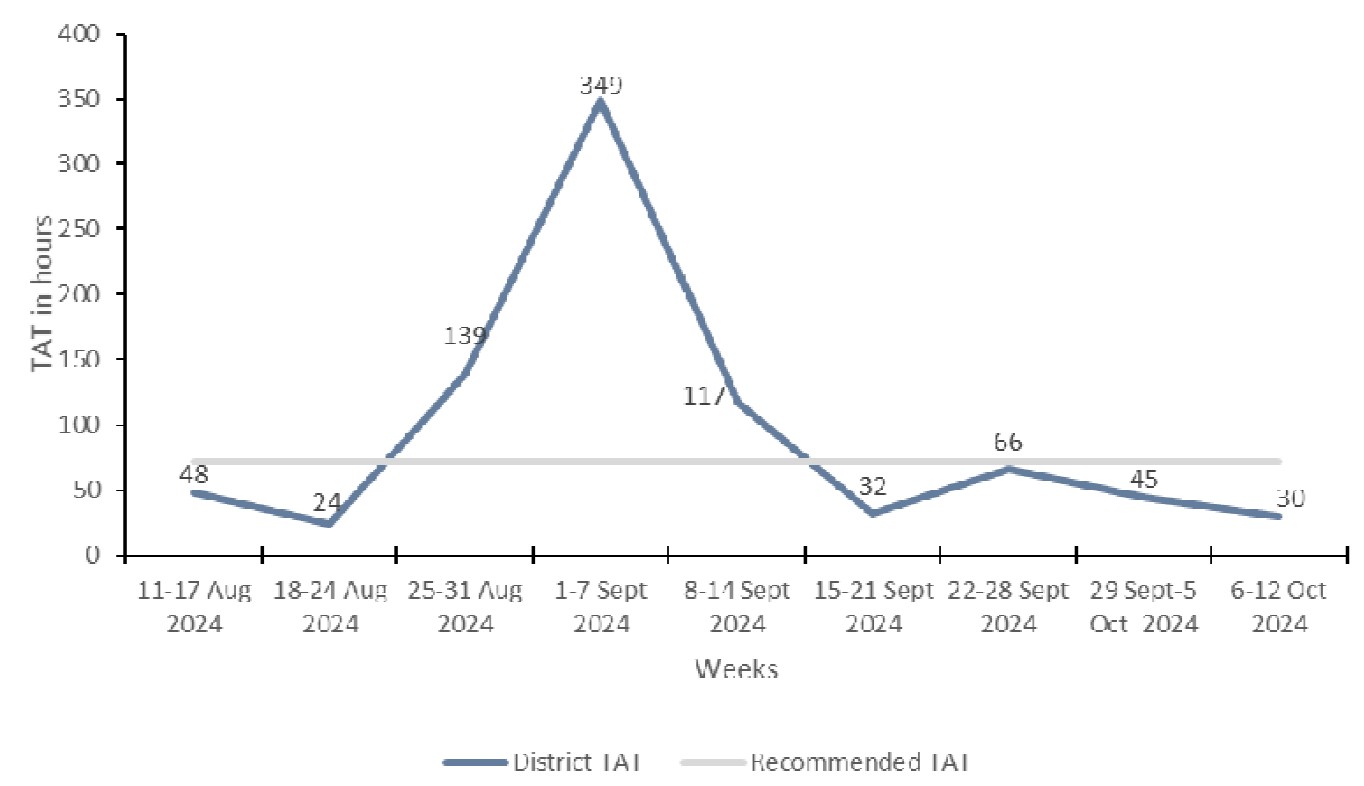
Background: On July 24, 2024, Uganda confirmed two mpox cases in Kasese District. The outbreak quickly spread to other districts including Mayuge. Rapid response teams were deployed to support the response (RRT). Despite interventions by the RRT, the affected districts faced challenges including long Turnaround Time (TAT) for mpox result receipt of over 120 hours instead of the recommended 72 hours. In response, surge teams across the different pillars were deployed to support district response. We describe the improvement of turnaround time (TAT) following the surge team deployment, Mayuge District, September–October 2024. Methods: Through meetings and brainstorming sessions, we worked with the district and health facility teams to map four health facilities where mpox cases were most likely to be identified. We constituted a district rapid response team and identified factors contributing to long TAT for result receipt, conducted root cause analysis using the 5-why technique, and implemented interventions in Plan-Do-Check-Act (PDCA) cycles selected based on feasibility and impact to address the root causes. Result receipt TAT was defined as the time from the date of sample collection to the date the results were received in hours. Data was abstracted from the sample referral register and result dispatch system (RDS). Results: The factors contributing to long mpox TAT for result receipt included inadequate supplies such as mpox request forms, Dacron swabs, gaps in sample collection and handling, including uncertainty about which lesions to sample and how to properly package specimens, and the lack of dedicated sample transport to hub and testing laboratories.To address these, we trained 33 health workers on Mpox sample collection and packaging, mobilized mpox supplies and activated the emergency sample transport network. Following the interventions, the TAT reduced from 118 hours (24-349 hours) on average to 56 hours (45-66 hours) on average in five weeks. Conclusion: The surge teams strengthened local laboratory systems by training, supply requisition and activation of the emergency sample transport network which reduced the TAT of mpox in Mayuge District. These interventions were essential for improving turnaround time.
Background
On July 24, 2024, Uganda confirmed two mpox cases in Kasese District (1). This led to activation of the response pillars including the laboratory pillar. According to the World Health Organization emergency response framework, the laboratory pillar works with other pillars to provide diagnostic support to detect and confirm Mpox cases that may arise among all suspects (2). In this response, the laboratory pillar worked with surveillance teams to identify priority areas and populations at risk and conduct investigations as well as support case management teams who care for identified cases. Interventions such as training both physical, and virtual were conducted at national and subnational level, activation of the result dispatch system and preposition of sample collection and management supplies were instituted to ensure rapid identification and control of the outbreak.
Despite the interventions, the outbreak quickly spread to other districts including Mayuge. Following the outbreak in these districts, challenges were reported during the national taskforce meetings from teams on ground responding to the outbreak. Among the challenges specific to the laboratory pillar was long mpox Turnaround Time (TAT) for result receipt. The results were reported to be taking over 120 hours instead of the recommended 72 hours. Being an emerging disease, the Ministry of Health dispatched surge teams across the different pillars to support districts. We describe the improvement of mpox turnaround time (TAT) following the surge team deployment, Mayuge District, September–October 2024.
Methods
Study setting and context: Mayuge District was among the mpox hotspot districts in Eastern Uganda with long TAT despite its proximity to the testing laboratories. We specifically focused response improvement in four health facilities that were critical to the Mpox response based on case volume, geographical location, and their capacity to handle Mpox-related activities.
Baseline assessment: We reviewed the referral register and downloaded Mayuge mpox data from the Result Dispatch system (RDS) for the month of August to establish the baseline TAT for the mpox result receipt. We defined TAT for result receipt as the time from the date of sample collection to the date the results were received in hours.
This was then compared against the TAT set by the national laboratory pillar which was 72 hours. Identification of factors contributing to long mpox result receipt turnaround time and root cause analysis
We formed a district rapid response team that included the laboratory managers, one laboratory technician from each facility, hub coordinator, and the district laboratory focal person (DLFP) to lead the TAT improvement initiative. We brainstormed to identify factors contributing to the long TAT.
These were listed and those that appeared the most were taken as the factors contributing to the long TAT. Root cause analysis was done using the 5-why technique. Interventions to improve result receipt turnaround time Following root cause analysis, interventions were prioritized based on impact and feasibility. Two PDCA cycles were implemented. The first cycle (8th-14 September 2024) involved training of health workers and provision of essential supplies.
The second cycle was implemented between 22nd and 28th September 2024 which involved deployment of standby vehicles and motorcycles to pick the samples as and when required. Intervention monitoring and evaluation and data analysis To monitor the effect of the interventions, weekly mpox data was downloaded from RDS and ab-This was plotted on a line graph to assess the trends.
Ethical considerations: The Ministry of Health Uganda provided administrative clearance to conduct this work as part of emergency response initiatives. In addition, we received a non-research determination clearance from the US Centers for Disease Prevention and Control (US CDC). This activity was reviewed by the CDC and was conducted consistent with applicable federal law and CDC policy. We sought verbal consent from the district and facility teams that we worked with to identify factors contributing to long TAT and implement interventions.
Results
Baseline mpox result receipt turnaround time, contributing factors, and root causes Following records review, the baseline TAT for the mpox samples referred from Mayuge through the hub was on average 118 hours. We identified nine factors that contributed to the long TAT and these included inadequate sample collection and packaging materials, unclear roles in sample and result management, poor communication between hub and facilities, limited supervision and accountability, inadequate staffing, absence of emergency protocols, unfavorable weather, knowledge gap in mpox sample collection as the teams did not know the best lesion to swab and the type of swab to be used. Some preferred to prick which was against the guidelines. The facilities also did not have dedicated sample transport means to ship samples from the facilities to the testing laboratories. They relied on the routine sample transport schedule where samples were picked twice or thrice a week. Using the 5-why technique, we identified three root causes for the long TAT, and these were knowledge gap on mpox sample collection and packaging, inadequate supplies and lack of dedicated sample transport for samples to the testing laboratories.
Effect of interventions on long result receipt turnaround time
To address the root causes, we implemented three interventions in two PDCA cycles. In the first PDCA cycle between 8th and 14th, Sep. We received and pre-positioned 200 swabs with Viral Transport Media (VTM), and four booklets of request forms. We trained 33 health workers on Mpox sample collection and packaging. They were trained using both didactic and practical sessions. The staff were trained on mpox sample collection and management, donning and doffing, waste management, sample tracking, requisition of stocked out supplies as well as result management. This reduced the TAT for one week to 32 hours from 117 hours. However, this change was not sustained as the TAT increased to 66 hours. The second PDCA cycle was implemented between 22nd and 28th September 2024. This involved activation of the emergency sample transport network by providing standby vehicles and motorcycles to pick the samples as and when required. This reduced the TAT to 30 hours (Figure1)
Discussion
The deployment of surge teams improved mpox result receipt TAT in Mayuge District. The root causes for the long TAT were knowledge gap on mpox sample collection and packaging, inadequate supplies and lack of dedicated sample transport for samples to the testing laboratories. These findings are consistent with the mpox intra action review gaps identified conducted by the Ministry of Health (3). When teams are trained, it boosts their confidence and increases their knowledge on sample and result management hence the reduced TAT. This is in agreement with Kangbai et al., 2025 where capacity building is one of the key recommended interventions in tackling The activation of the emergency sample transportation system played a crucial role in reducing the turnaround time for Mpox sample testing.
Swift transport ensured timely diagnosis and case management, demonstrating the importance of a robust transport network for disease control. This led to the reduction of TAT as samples were transported to the testing laboratories as and when collected. This agrees with the Africa CDC initiative of ensuring samples are timely transported for effective response (5). Continuous monitoring of stock levels, prompt requisition of materials, and collaboration with partners are necessary to maintain an uninterrupted supply chain, especially in high-demand situations which sometimes leads to long TAT as districts and facilities have to borrow from their counterparts.
Training and activation of the emergency sample transport network together with ensuring supply availability improved the TAT.
Study limitations: Being an outbreak response, the study period was short (five weeks), making it difficult to assess the sustainability of the interventions. We also did the study in one district which may constrain generalizability. The study focused primarily on turnaround time and did not directly measure the impact of the interventions on the response in general, thereby limiting the ability to establish a direct correlation between TAT and effective surveillance and case management.
Conclusion
Long TAT was caused by inadequate mpox supply availability, knowledge gap in sample collection and management as well as lack of stand by transport for sample transportation to the testing laboratories. The surge teams strengthened local laboratory systems by training, supply requisition and activation of the emergency sample transport network. These interventions improved the turnaround time for mpox testing in Mayuge District.
Conflict of interest: The authors declare that they had no conflict of interest.
Authors contribution: We designed the study and data analysis with guidance from GS, SN and RK. YK, WO, RI, TR, RN and WA did the data collection. All authors agreed to the submission of the article.


Comments are closed.