Strengthening Management of Non-Conformities in an Accredited Public Health Laboratory in Kampala, Uganda, March – October 2024
Authors: Ritah Namusoosa1,2*, Samuel Gidudu1, Eunice Jennifer Nambozo2, Nalwanga Rebecca2, Esther Nabende2, William Senyonga2, Martha Pedun3, Grace Najjuka2 Institutional affiliations:1Uganda Public Health Fellowship Program-Laboratory Leadership Program; Uganda National Institute of Public Health; Ministry of Health Kampala, Uganda; 2National Microbiology Reference Laboratory, National Health Laboratory and Diagnostics Services; Ministry of Health, Kampala, Uganda, 3African Society for Laboratory Medicine; Kampala, Uganda. Correspondence*: Tel: +256785842878, Email: rnamusoosa@uniph.go.ug
Summary
Background: The management of non-conformities (NC) is a core International Organization for Standardization (ISO)15189 requirement for maintaining medical laboratory accreditation because it provides a structured process for identifying, addressing, and preventing deviations, thereby supporting a risk-based quality management system that ensures patient safety, reliable results, and continual improvement. In March 2023, a College of American Pathologist (CAP)-accredited public health laboratory in Uganda showed suboptimal NC management, with an average performance of 38% (48% documentation; 27% investigation/closure), revealing major quality gaps. We set out to identify barriers to optimal NC management and improve the average NC management performance rate to 80% by September 2024.
Method: We conducted two Plan-Do-Check-Act (PDCA) cycles, with performance compared against a 12-month retrospective baseline (January 2023–January 2024) and monitored prospectively on a monthly basis during and after each intervention.”. Barriers were identified through brainstorming sessions and prioritized using Pareto analysis. Root causes were determined using the 5-why technique, which identified an unclear job description (JD) for the NC focal person and the lack of a regular review schedule as the primary root causes. Interventions included reviewing and clarifying the NC focal person’s JD, and implementing a mandatory biweekly NC review and follow-up schedule.
Results: Following the interventions, the average NC management performance rate increased from 38% to 100%. Both the documentation rate and the investigation/closure rate improved to 80% by October 2024, and successfully exceeding the project’s 80% goal while observing its sustainability.
Conclusion: Strengthening roles and introducing biweekly reviews effectively restored the NC management system. To ensure the sustainability of this progress, we recommend the maintenance of regular reviews and the periodic updating of NC management responsibilities.
Background
A core International Organization for Standardization (ISO) 15189 requirements is systematic non-conformity (NC) management, which includes documenting deviations, investigating root causes, implementing corrective actions, and verifying their effectiveness to prevent recurrence (1–4). This requirement is central to accreditation of health laboratories. Accreditation of health laboratories is fundamental for ensuring the accuracy, reliability, and timeliness of patient results and strengthening overall health systems. The College of American Pathologists (CAP) accreditation, guided by international standards like ISO 15 189, validates a laboratory’s competence and reinforces a culture of continuous quality improvement (1,2). Effective NC management therefore reinforces continuous quality improvement within the Quality Management System.
The NCs may arise across the pre-analytical, analytical, and post-analytical phases and are commonly detected through internal audits, staff observations, customer complaints, or routine process monitoring (3,4). However, many laboratories continue to face persistent challenges in NC management, including weak root cause analysis, delayed implementation of corrective and preventive actions (CAPA), poor documentation practices, and limited staff engagement. These gaps contribute to recurring errors, threaten accreditation status, and compromise the overall quality and reliability of laboratory services (5,6).
In March 2024, an internal review at a CAP-accredited public health laboratory in Kampala, Uganda revealed significant weaknesses in NC management. Of the 31 NCs identified in 2023, only 48% were formally documented and just 27% of those documented had been investigated and closed, resulting in an average performance of 38%. Several undocumented NCs were classified as major, posing direct risks to patient care through delays in turnaround time, repeated QC failures, and equipment breakdowns. These findings highlighted systemic issues within the Quality Management System and underscored the need for targeted intervention. To address these gaps, we initiated a Continuous Quality Improvement (CQI) project aimed at improving NC management performance from 38% to 80% by September 2024.
Method
Setting and context: This CQI project was conducted from March to September 2024 at a CAP-accredited public health laboratory in Kampala, Uganda, operating under ISO 15189 and CAP standards. The project targeted improvement of NC management processes within the laboratory quality management system, including identification, documentation, investigation, corrective action and preventive action (CAPA) implementation, and closure.
Continuous quality improvement approach: The objectives were to identify barriers to optimal NC management and to improve the average NC management performance rate to 80% by September 2024. A multidisciplinary team applied the Plan-Do-Check-Act (PDCA) model to guide iterative improvements. A retrospective review of QMS records (January 2023–January 2024) established baseline performance (38%). Barriers were identified through brainstorming with 22 laboratory staff and prioritized using Pareto analysis, and root causes were determined using the Five Whys technique.
Iterative improvement cycles: Two PDCA cycles were implemented. Cycle 1 (April–May 2024) clarified the NC focal person’s job description, resulting in modest improvement in documentation. Cycle 2 (June–September 2024) introduced mandatory biweekly NC review meetings, leading to substantial and sustained improvements in both documentation and closure. The biweekly review process was institutionalized through an SOP to support sustainability.
Measures and data analysis: Performance was monitored using two indicators: the proportion of documented NCs and the proportion of investigated and closed NCs. An average NC management performance rate was calculated biweekly and displayed on a run chart to assess trends and the impact of each PDCA cycle over time.
Ethical considerations: We sought a non-research determination from the Associate Director for Science of the US Centres for Disease Control and Prevention. This project was classified as a quality improvement initiative as it used only routinely collected, anonymized operational data for analysis. We also sought administrative clearance from the management of the laboratory to conduct the project. This project was conducted consistent with applicable federal law and the US CDC policy. §§See e.g., 45 C.F.R. part 46, 21 C.F.R. part 56; 42 U.S.C. §241(d); 5 U.S.C. §552a; 44 U.S.C. §3501 et seq.
Results
Baseline performance and prioritized barriers
At baseline (January 2023–2024), NC management performance was low. Of the 31 NCs identified, only 48% (15/31) were documented and 27% (4/15) were fully investigated and closed, resulting in an average performance rate of 38% (Figure 1). Four our major barriers to NC management were identified: irregular NC review and follow-up, lack of accessible NC documentation forms, an unclear job description for the NC focal person, and absence of a centralized NC filing system, which together accounted for approximately 80% of performance gaps (Figure2). And the main root causes were an unclear job description for the NC focal person and the absence of a structured schedule for routine NC review and follow-up.
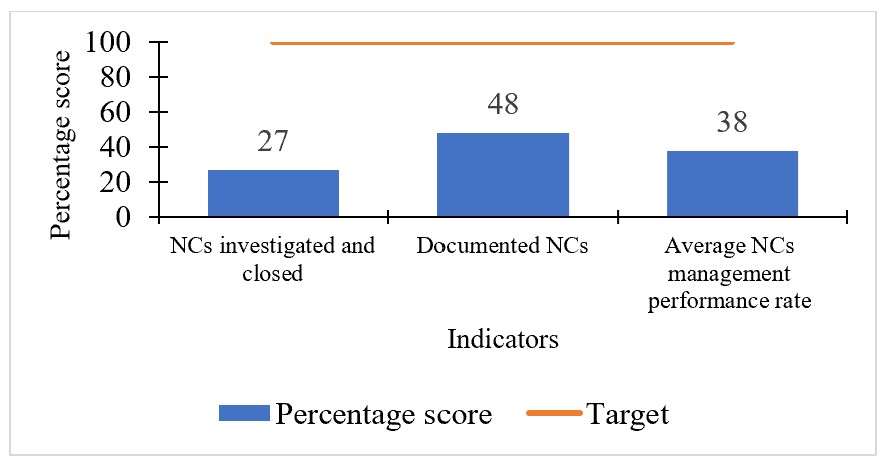
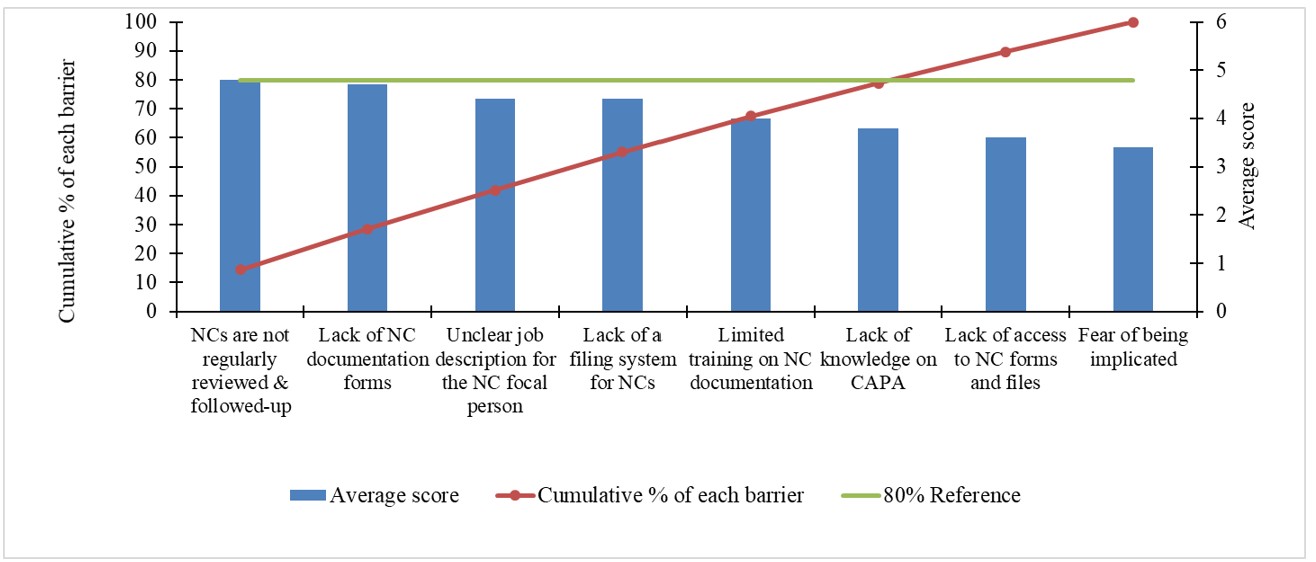
non conformitiess in the laboratory between January 2023 between January 2024
Improvement in Non-conformity management over time
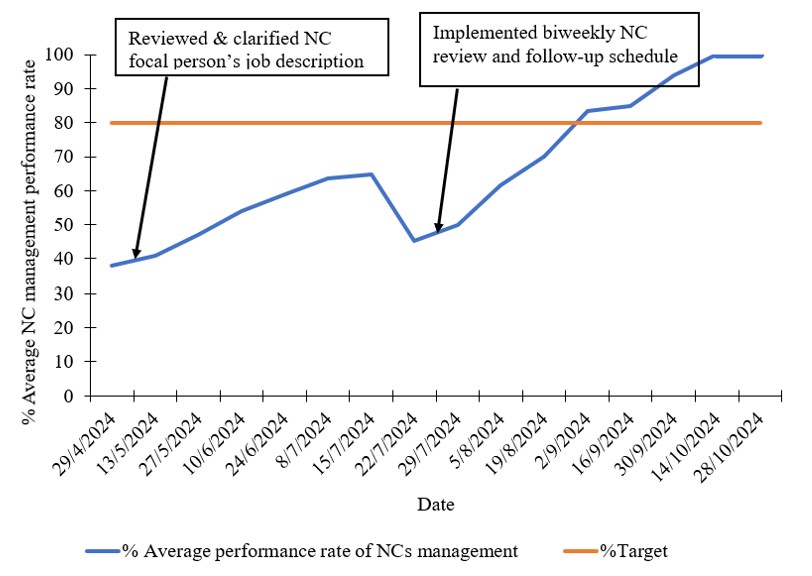
Following implementation of two PDCA cycles, NC management performance improved substantially (Figure 3).
Following the implementation of the two PDCA cycles, the average NC Management Performance Rate improved (Figure 3).
After clarification of the NC focal person’s job description, the performance rate increased 38% to 65%. A temporary decline to 52% was observed in July, followed by sustained improvement after introduction of mandatory biweekly NC review meetings. The project’s 80% target attained in September 2024, and rose to 100% in October 2024.
Discussion
This CQI project improved NC management performance from 38% to 100%, with documentation and investigation/closure rates exceeding the 80% target. Key contributing factors included clarification of the NC focal person’s job description and the introduction of biweekly NC review meetings, which strengthened accountability and alignment with ISO 15189 and CAP requirements, while continuous quality improvement.
Initial improvements were attributable to enhanced NC documentation, whereas sustained performance improvement required structured review mechanisms to ensure timely investigation and closure, reflecting progressive strengthening of the NC management process. These findings are consistent with reports from LMICs, including Strengthening Laboratory Management Toward Accreditation (SLMTA) implementation in Tanzania and Stepwise Laboratory Quality Improvement Process Towards Accreditation (SLIPTA) experiences in Kenya, where structured CQI approaches have significantly improved laboratory quality management systems (7,8).
The PDCA-based framework: defining roles and review schedules (Plan), implementing corrective actions (Do), monitoring performance indicators (Check), and institutionalizing effective practices (Act); is well supported in the literature. The temporary performance decline observed during implementation mirrors fluctuations reported in other CQI initiatives and highlights the importance of sustained monitoring and reinforcement (9,10). Overall, the findings reaffirm that robust documentation systems, clearly defined accountability structures, and routine follow-up are essential for effective NC management and long-term accreditation sustainability in resource-limited settings (1,10).
Study limitations
The evaluation period was relatively short (six months), making it difficult to assess the long-term sustainability of the interventions. The study also relied on routinely collected QMS data and a single-site design, which may constrain generalizability. It also focused primarily on process indicators (proportion of NCs documented, investigated, and closed) and did not directly measure the impact on patient care, thereby limiting the ability to establish a direct correlation between the process improvements and the ultimate quality of laboratory service.
Conclusion
This project showed that a weak NC management system can be fully restored by systematically addressing its root causes. To ensure the sustainability of this progress, we recommend the maintenance of regular reviews and the periodic updating of NC management responsibilities.
Conflict of interest: The authors declare no conflict of interest.
Authors Contribution: RN: conceptualized, designed, and contributed to data analysis; EJN, NR, EN, WS and GN: participated in data collection and cleaning; RN: led the writing of the bulletin; SG, and MP, participated in bulletin writing and review to ensure intellectual content and scientific integrity. All authors read and approved the final bulletin.
Acknowledgments: We acknowledge the Uganda Public Health Fellowship Program, National Microbiology Reference Laboratory (NMRL), National Health Laboratory and Diagnostic Services, Ministry of Health Uganda, Makerere University School of Public Health and Baylor Uganda for the technical support and oversight of this project. We also acknowledge the US Centres for Disease Control and Prevention Uganda for implementation support.
Copyright and licensing: All material in the Uganda Public Health Bulletin is in the public domain and may be used and reprinted without permission. However, citation as to source is appreciated. Any article can be reprinted or published. If cited a reprint, it should be in the original form.
References
- Tesema M, Sisay A. Medical laboratory accreditation status and associated factors in selected private and government health facilities of Addis Ababa, Ethiopia. Pan Afr Med J. 2023;45.
- Desalegn DM, Taddese BD, Yemanebrhane N, Getahun MS, Kitila KT, Dinku TT, et al. Medical laboratory accreditation in a resource-limited district health centre laboratory, Addis Ababa, Ethiopia. Afr J Lab Med. 2019;8(1):1–5.
- Youssef EH, Hafsa LA, Najat M, Leila R, Mimoun Z, Abdelkarim FM. Risk analysis and management of non-conformities of the pre-analytical phase in a university testing laboratory of bacteriology. J Med Lab Diagnosis. 2014;5(1):1–10.
- APHL. Nonconforming Events. 2021;(October).
- Tsheola E, Pienaar J, Kruger W. Analysis of internal audit non-conformances at non-accredited public health laboratories in Gauteng province, South Africa. J Med Lab Sci Technol South Africa. 2024;6(2):52–7.
- Mesganaw B, Fenta A, Hibstu Z, Belew H, Misganaw K, Belayneh M. Medical Laboratories Quality Management and Challenges in Ethiopia: A Systematic Review. Pathol Lab Med Int. 2023;Volume 15(February):13–26.
- Makokha EP, Ondondo RO, Kimani DK, Gachuki T, Basiye F, Njeru M, et al. Enhancing accreditation outcomes for medical laboratories on the Strengthening Laboratory Management Toward Accreditation programme in Kenya via a rapid results initiative. Afr J Lab Med. 2022;11(1):1–8.
- Andiric LR, Massambu CG. Laboratory Quality Improvement in Tanzania. Am J Clin Pathol [Internet]. 2015 Apr 1;143(4):566–72. Available from: https://doi.org/10.1309/AJCPAB4A6WWPYIEN
- Endalamaw A, Khatri RB, Mengistu TS, Erku D, Wolka E, Zewdie A, et al. A scoping review of continuous quality improvement in healthcare system: conceptualization, models and tools, barriers and facilitators, and impact. BMC Health Serv Res [Internet]. 2024;24(1):1–14. Available from: https://doi.org/10.1186/s12913-024-10828-0
- Hill J, Stephani A, Sapple P, Clegg A. The effectiveness of continuous quality improvement for developing professional practice and improving health care outcomes: A systematic review. Implementation Science [revista en Internet] 2020 [acceso 14 de junio de 2024]; 15(1): 1-14. Implement Sci [Internet]. 2020;15(23):1–14. Available from: https://implementationscience.biomedcentral.com/articles/10.1186/s13012-020-0975-2


Comments are closed.