Temporal and Spatial Distribution of Human Brucellosis, Uganda, 2017–2024
Authors: Aminah Namwabira1*, Justine Wobusobozi1, Martha Dorcas Nalweyiso1, David Muwanguzi2, Stella Maris Lunkuse2, John Opolot2, Alfred Weguli2, Benon Kwesiga1, Richard Migisha1, Alex Riolexus Rio1 Institutional Affiliations: 1Uganda Public Health Fellowship Program, Uganda National Institute of Public Health, Kampala, Uganda, 2Ministry of Health, Department of Integrated Epidemiology, Surveillance and Public Health Emergencies, Division of Veterinary Public Health and Zoonoses, Kampala, Uganda *Correspondence: Email: anamwabira@uniph.go.ug , Tel +256771894690
Summary
Background: Brucellosis is a highly infectious zoonotic bacterial infection, leading to a chronic illness in humans. Despite its zoonotic and economic importance, limited information on brucellosis continues to hinder timely implementation of effective interventions. We analyzed surveillance data to describe the temporal and spatial distribution of human brucellosis, Uganda, 2017–2024.
Methods: We abstracted surveillance data on reported annual cumulative clinically diagnosed brucellosis cases from the District Health Information Software 2 for the period of 2017–2024. A brucellosis case was acute or insidious onset of fever and ≥ one of the following: night sweats, arthralgia, headache, anorexia, myalgia, weight loss, arthritis, meningitis or focal organ involvement. We calculated annual and median incidence rates, assessed significance using Mann-Kendall trend test.
Results: A total of 1,542,564 cases were reported during 2017–2024 at 90% reporting rate. Overall median incidence rate was 115/100,000 (IQR=88/100,000-194/100,000) with no change in the trend over the years (p =0.10). Brucellosis cases increased significantly in Health Centre III (p=0.019, gradient=1.8). Karamoja Region was the most affected in all the years with a median incidence rate of 733/100,000 (IQR=650/100,000-854/100,000).
Conclusion: The findings indicate a consistently high burden of reported brucellosis cases over the eight-year period with no significant change in the trend, suggesting stability of the disease during 2017–2024. Only Health Centre III registered an increase in the proportion of brucellosis cases while Karamoja Region was the most affected throughout the study period. Conducting community-based education and behavior change programs could reduce transmission of brucellosis to humans.
Background
Brucellosis is a highly infectious zoonotic bacterial infection caused by several species of the genus Brucella. Brucellosis occurs globally with an estimate 1.6 to 2.1 million new human cases per year and Africa contributes 0.5 million cases annually to the global burden(1). Brucellosis causes a severe debilitating disease in people and a significant economic impact in livestock (2). Brucellosis is primarily transmitted to humans through direct contact with infected animals or consumption of contaminated animal products, including unpasteurized dairy products and uncooked meat (3). Brucellosis is considered an occupational hazard for people who work with animals, including veterinarians, animal owners, herdsmen, abattoir workers, butchers and laboratory personnel, as they handle infected animals, aborted fetuses and placentas (4). Brucellosis is endemic in Uganda, with a national human sero-prevalence of 17% (5). Routine analysis of surveillance data for an endemic disease like brucellosis is essential for early detection of outbreaks, identification of high-risk populations, and effective implementation of public health interventions. However, despite its importance, information on temporal trends and spatial distribution of brucellosis remains limited, hindering timely outbreak detection and response. We analyzed surveillance data to describe the temporal and spatial distribution of human brucellosis, Uganda, 2017–2024.
Methods
We conducted a retrospective descriptive analysis using brucellosis surveillance data from the Ministry of Health (MoH) District Health Information Software 2 (DHIS2).
Data on reported annual cumulative brucellosis cases, reporting health regions, health facility level and reporting rates were extracted from DHIS2 monthly report HMIS 105 for the period of 2017–2024 and managed using Microsoft Excel. As per DHIS2, a brucellosis case was defined as acute or insidious onset of fever and ≥ one of the following: night sweats, arthralgia, headache, anorexia, myalgia, weight loss, arthritis, meningitis or focal organ involvement. We obtained data on mid-year projected populations for 2017–2024 from the Uganda Bureau of Statistics (UBOS).
We calculated the annual incidence rate of brucellosis using the number of reported cases as the numerator and the UBOS mid-year projected population as the denominator. We used Microsoft Excel to generate line graphs showing temporal trends over eight years. Trends were tested for significance using the Mann–Kendall test in R Studio (p < 0.05), and the trend slope (gradient) was also obtained in R. We calculated the annual percentage change in incidence rate as the gradient divided by the median incidence rate, multiplied by 100. The median was used because the data were not normally distributed. Choropleth maps showing regions with the highest annual incidence rates were generated in Quantum Geographic Information System.
The descriptive study was conducted using routine surveillance data collected by Ministry of Health as part of ongoing disease monitoring and control activities. Authorization to access and analyze the data was granted from Ministry of Health. A non-research determination was obtained from the office of the Associate Director for Science, Centres for Disease Control and Prevention, Uganda. Data collected in DHIS2 is aggregate without individual patient data identifiers.
Results
Temporal distribution of human brucellosis, Uganda, 2017–2024
A total of 1,542,564 brucellosis cases were reported for the eight years reviewed. The average reporting rate over the study period was 90%. The highest brucellosis incidence rate (430/100,000) was registered in 2019 while the median incidence rate was 115/100,000 (IQR=88/100,000-194/100,000) with no change in the trend over the years (p =0.10) (Figure 1).
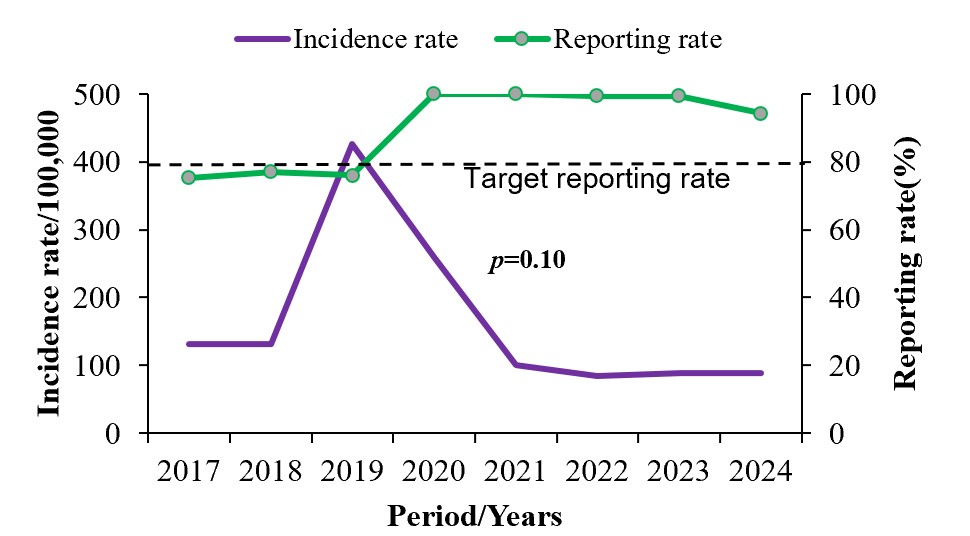
Figure 1: Temporal distribution of human brucellosis in Uganda, 2017–2024
Trends of human brucellosis by health facility level, Uganda, 2017–2024
The proportion of brucellosis cases increased significantly in Health Centre III (HCIII)
(p=0.019, gradient=1.8) from 31% (15,278/48,884) in 2017 to 47% (18,572/39,286) in 2024 (Figure 2).
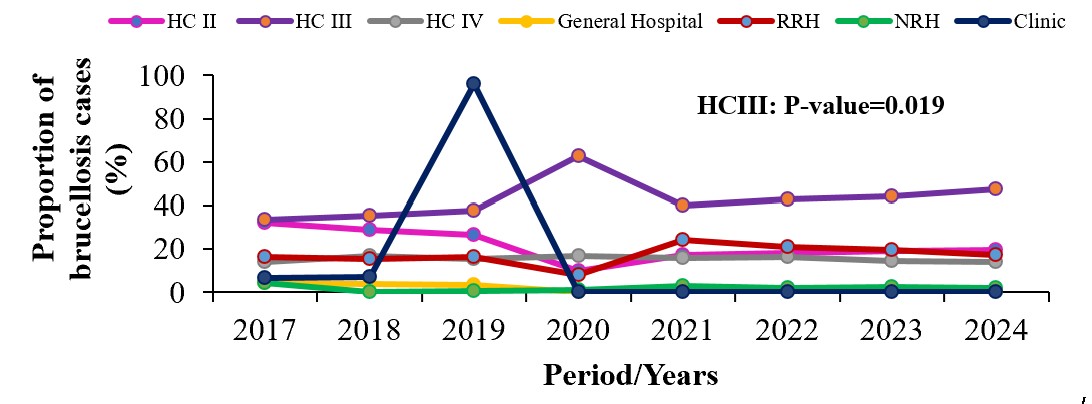
Figure 2: Trends of human brucellosis by health facility level, Uganda, 2017–2024
Spatial distribution of human brucellosis incidence by regions and districts, Uganda, 2017–2024
Karamoja region consistently had the highest incidence rate throughout 2017 to 2024 with a median incidence rate of 733/100,000 (IQR=650/100,000-854/100,000)
(Figure 3).
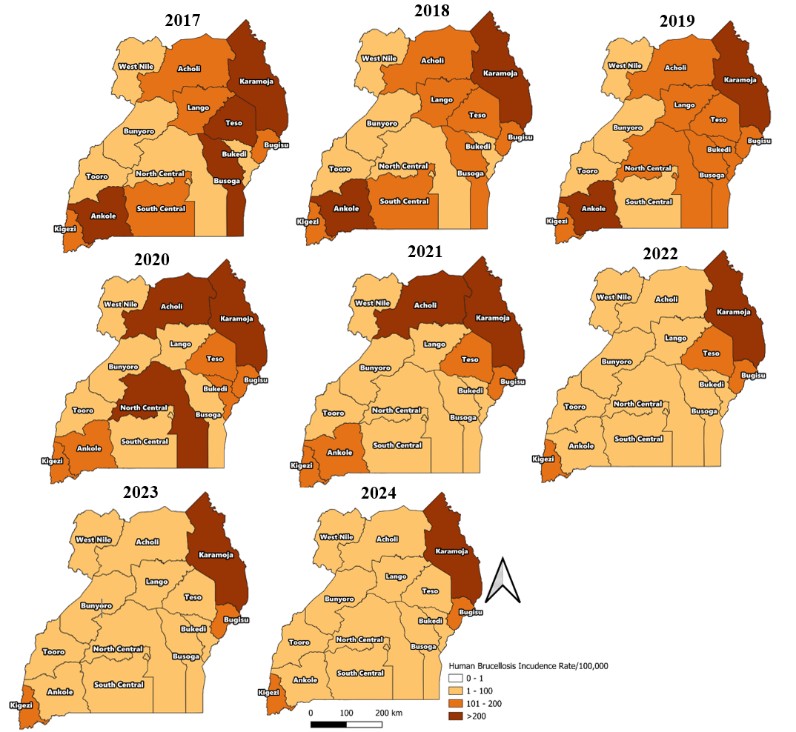
Figure 3: Spatial distribution of human brucellosis incidence by region, Uganda, 2017–2024
Discussion
Over the eight years’ period analyzed, a total of 1,542,564 brucellosis cases were reported, at an average reporting rate of 90%, reflecting a substantial public health burden. The annual global incidence of brucellosis estimated by Centers for Disease Control in 2023 was 2.1 million (1). With 1,542,564 cases in eight years, Uganda contributes 9.2% to the total global incidence of reported brucellosis cases every year. This finding indicates a persistently high incidence signifying that brucellosis is a major endemic zoonosis in the country (6).
The proportion of brucellosis cases increased significantly in Health Centre III (HCIII)
from 2017 to 2024. Health Centre IIIs have better equipment, supplies, and workforce compared to Health Centre IIs, they are more accessible to rural communities than HCIV, General Hospitals, RRH or NRH and are cheaper compared to clinics (7) .This is likely to make Health Centre III a preference as a first point of care for brucellosis patients, reflecting the significant increase in the proportion of cases reported.
Karamoja region consistently had the highest incidence rate of brucellosis throughout 2017 to 2024. Majority of the population in Karamoja region are nomadic pastoralists who live in close contact with livestock and engage in cultural practices such as consumption of unboiled milk, raw meat and raw blood which increase their risk of exposure to brucellosis (8).
Study limitations
We analyzed clinically diagnosed brucellosis cases because of a small number of laboratory-confirmed brucellosis cases in DHIS2. The small number could not allow for meaningful statistical analysis and would lead to non-representative of the true disease burden. Consequently, this study describes the temporal and spatial distribution of reported human brucellosis cases in Uganda and does not estimate the true disease burden.
Conclusion
The findings indicate a consistently high burden of reported brucellosis cases over the eight-year period with no significant change in the trend, suggesting stability of the disease during 2017–2024. Only Health Centre III registered an increase in the proportion of brucellosis cases while Karamoja region persistently had the highest incidence of brucellosis throughout the study period. We recommend conducting community-based education and behavior change programs to reduce the risk of transmission of brucellosis to humans. We further recommend establishing data sharing platforms between Ministry of Health, Ministry of Agriculture, Animal Industry and Fisheries and the local governments to monitor trends and outbreaks in real time.
Conflict of Interest
The authors declare no conflict of interest
Author contribution
Aminah Namwabira took lead in conceptualizing the project, data abstraction, analysis and original draft writing. Justine Wobusobozi, Martha Dorcas Nalweyiso, Benon Kwesiga and Richard Migisha were involved in designing the methodology, reviewing and editing the article. Stella Maris Lunkuse, David Muwanguzi, Alfred Wejuli, John Opolot and Alex Riolexus Ario were involved in supervision, visualization, validation and editing the article. All authors read and approved the final draft.
Acknowledgements
The authors appreciate the Ministry of Health for providing access to DHIS2 data that was used for this analysis. We further appreciate the Department of Integrated Epidemiology, Surveillance and Public Health Emergencies, the Division of Veterinary Public Health and Zoonoses for raising the concerns that initiated this analysis.
Copyrighting and licensing
All material in the Uganda Public Health Bulletin is in the public domain and may be used and printed without permission. However, citation as to source is appreciated. Any article can be reprinted or published. If cited as a reprint, it should be referenced in the original form.
References
- Laine CG, Johnson VE, Scott HM, Arenas-Gamboa AM. Global Estimate of Human Brucellosis Incidence. Emerg Infect Dis [Internet]. 2023 Sep 1 [cited 2025 Nov 22];29(9):1789. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC10461652/
- Brucellosis in humans–etiology, diagnostics, clinical forms – PubMed [Internet]. [cited 2025 Nov 22]. Available from: https://pubmed.ncbi.nlm.nih.gov/23772567/
- Clinical Overview of Brucellosis | Brucellosis | CDC [Internet]. [cited 2025 Nov 22]. Available from: https://www.cdc.gov/brucellosis/hcp/clinical-overview/index.html
- World Health Organization. Brucellosis [Internet]. 2020 [cited 2025 Nov 22]. Available from: https://www.who.int/news-room/fact-sheets/detail/brucellosis
- Patricia Eyu, Edrida M, Tukahebwa, Benon Kwesiga ARA. UNIPH Bulletin Articles Volume 2. 2017 [cited 2025 Nov 21]. Analysis of Surveillance data to determine distribution of Human Brucellosis in Uganda, July 2015 to June 2017 – UNIPH. Available from: https://uniph.go.ug/analysis-of-surveillance-data-to-determine-distribution-of-human-brucellosis-in-uganda-july-2015-to-june-2017/
- Djangwani J, Ooko Abong’ G, Gicuku Njue L, Kaindi DWM. Brucellosis: Prevalence with reference to East African community countries – A rapid review. Vet Med Sci [Internet]. 2021 May 1 [cited 2025 Nov 22];7(3):851–67. Available from: https://pubmed.ncbi.nlm.nih.gov/33421354/
- Ssempiira J, Kasirye I, Kissa J, Nambuusi B, Mukooyo E, Opigo J, et al. Measuring health facility readiness and its effects on severe malaria outcomes in Uganda. Sci Reports 2018 81 [Internet]. 2018 Dec 18 [cited 2025 Nov 22];8(1):17928-. Available from: https://www.nature.com/articles/s41598-018-36249-8
- Akwongo CJ, Kakooza S. Exposure to Brucella spp. in Goats and Sheep in Karenga District, Uganda Diagnosed by Modified Rose Bengal Method. Zoonotic Dis. 2022;2(3):163–71.


Comments are closed.